Summary of the study
To advance research and improve the management of resistant hypertension: participate in the clinical trial
To advance research and improve the management of resistant hypertension: participate in the clinical trial

« Medico-economic Evaluation of Unilateral Carotid Sinus Stimulation in the Treatment of Resistant Hypertension: A Randomized, Open-label, Multicenter, Controlled Trial with Safety and Blood Pressure Efficacy Evaluation (IDRCB : 2014-A00632-45) ».
You are under 85 years of age,
You are currently being monitored for high blood pressure,
Your blood pressure remains high (> 140 mmHg systolic blood pressure and/or > 90 mmHg diastolic blood pressure) despite taking at least three antihypertensive medications,
The NANCY CHRU and several Centres of Excellence in Hypertension of the French Hypertension Society offer you to participate in the ESTIM-rHTN clinical trial promoted by the Nancy CHRU and approved by the health authorities.
This study supported by the Ministry of Health aims to evaluate the efficacy and safety of an implantable medical device Barostim neo™ in high blood pressure compared to usual management.
This study will also assess the place and interest that the implantable medical device Barostim neo™ can take compared to an exclusive drug treatment with regard to the management of this disease already existing in France.
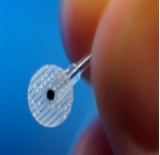
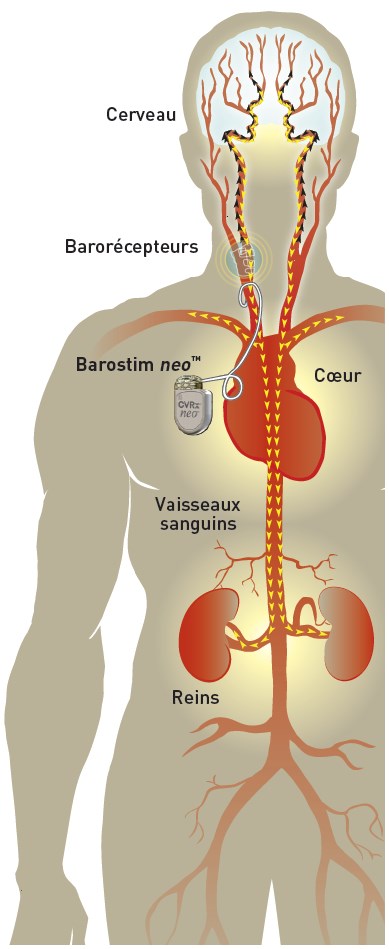

The Barostim system neo™ is an implantable device with CE marking designed to stimulate the body’s natural cardiovascular regulators called « baroreceptors » (blood pressure receptors) thus lowering blood pressure.
It consists of placing a small pellet (electrode) connected to a battery (the barostimulator) at an artery in the neck that is placed under the skin; it is called an implantable device. So it’s a surgical treatment and it’s not a drug.
The pacermaker resembles pacers that are commonly used to treat certain heart diseases.
The surgical procedure is of short duration; it is similar to the insertion of a cardiac pacemaker and is reversible, i.e. it can be removed or deactivated at any time for any reason.
This study is conducted in two groups of patients with poorly controlled hypertension despite taking antihypertensive drugs: a group treated with the usual antihypertensive treatments according to current recommendations and a group treated with the medical device Barostim neo™ in addition to the usual antihypertensive treatments. The group’s allocation is drawn by lot (i.e. the study group to which you will belong will not depend on you or the study doctor, but will be pre-established by computerised drawing of lots). You will have a specialized follow-up in a Centre participating in the study (7 visits maximum) during 12 months)
Cardiology and HTA Unit
Prof Philippe GOSSE
Contact : Mme Julie GAUDISSARD
Cardiology and Cardiovascular Rehabilitation Unit
Dr Thierry DENOLLE
Mail : estim-dinard@cic-p-nancy.fr
Department of Vascular Medicine and HTA
Dr Pascal DELSART
By postal letter to the attention of Dr DELSART:
Service de Médecine vasculaire et HTA
Institut Cœur-Poumon, CHRU Lille
Bd Pr Leclercq, 59037 Lille Cedex
or mail : estim-lille@cic-p-nancy.fr
Investigation Clinical Center Pierre Drouin
Prof Patrick ROSSIGNOL
Tél : +33 3 83 15 73 05
Mail : estim-nancy@cic-p-nancy.fr
Cardiovascular Prevention Unit
Prof Xavier GIRERD
Consultation by appointment at +33 1 42 17 78 32 or +33 1 42 17 57 74
Mail : Sylvie.maison@aphp.fr
Department of hypertension, renal and cardiovascular diseases
Prof Michel AZIZI
Site internet : www.centre-hypertension.org
Diagnostic and Therapeutic Centre
Prof Jacques BLACHER
Mme Veronique LABRO
Tél : +33 1 42 34 80 25
Investigation Clinical Center 1402
Prof Samy HADJADJ
Tél : +33 5 49 44 46 89
Department of Internal Medicine and High Blood Pressure
Prof Béatrice DULY – BOUHANICK
Mail : amar-j.sec@chu-toulouse.fr
High blood pressure, risk factors and heart failure unit
Prof Atul PATHAK
Mail : apathak@clinique-pasteur.com
Nephrology-HTA, Dialysis, Transplantation Department
Prof Jean Michel HALIMI
Mail : estim-tours@cic-p-nancy.fr
Clinical research (involving the human person) is conducted in strict compliance with ethics and current legislation.
This research is funded by the Ministry of Health (Medico-economic Research Programme 2013 (PRME-13-0260))